Russians are known for the best vodka globally, but they might have to quit for two months straight. As per reports, Russians warned to avoid alcohol for at least six to eight weeks from the time they receive Sputnik V.
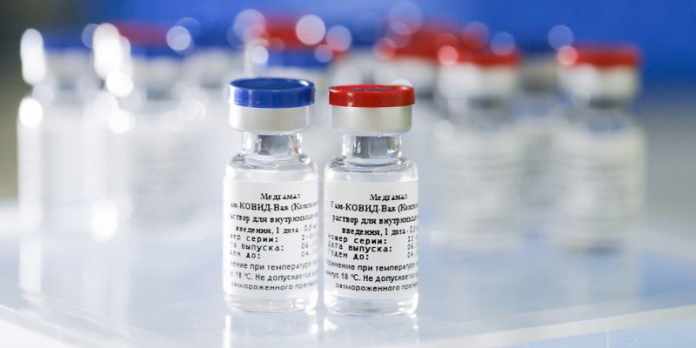
Sputnik V is the first claimed coronavirus vaccine manufactured in Russia. Many citizens are taking the vaccines to keep themselves safe from COVID. However, everyone has been warned to keep away from alcoholic beverages for two months straight.
Russians warned to avoid alcohol for 42 days
Tatiana Golikova, the Deputy Prime Minister, informed TASS that people need to keep away from hard drinks for at least 42 days since they vaccinate. They also have to quit immunosuppressant drugs to follow mitigation measures and avoid infection. Moreover, their medical community suggests that drinking excessively can weaken our immune system.
According to NIH, the consumption of excessive alcohol leads to immunity issues. People who drink a lot are more susceptible to pneumonia. Moreover, they have can other respiratory issues for the same reason.
In 2010, the average level of alcohol consumption was 15 liters or 4 gallons. In 2016, the numbers dropped to 11.7 liters or 3 gallons; WHO revealed the results in 2018. However, there is a high amount of episodic drinking among Russians. They are either more than or equal to 60% of present drinkers. WHO suggests that heavy alcohol consumption leads to liver cirrhosis, cancer, traffic-based injuries, and more.
Mass scale immunization ordered in Russia
President Vladimir Putin recently ordered a mass-scale immunization campaign of the COVID-19 vaccine. They are prioritizing doctors, essential workers, and teachers.
Already 100,000 residents took Sputnik V vaccine in Russia. However, experts across the globe have criticized the vaccine’s approval at a short span of time and with only dozens of volunteers.
In November, the vaccine developers said that the interim analysis of their trials showed 91.4% efficacy. The results were based on 39 infections among 18,794 volunteers who received two doses of either the vaccine or the placebo. The number of studies is much less compared to western drug-makers.


![Kyla Pratt Net Worth, Early Life, Career [2023] Kyla Pratt Net Worth](https://lessconf.com/wp-content/uploads/2023/06/Kyla-Pratt-Net-Worth-218x150.jpg)



